-
Courses

Courses
Choosing a course is one of the most important decisions you'll ever make! View our courses and see what our students and lecturers have to say about the courses you are interested in at the links below.
-
University Life

University Life
Each year more than 4,000 choose University of Galway as their University of choice. Find out what life at University of Galway is all about here.
-
About University of Galway

About University of Galway
Since 1845, University of Galway has been sharing the highest quality teaching and research with Ireland and the world. Find out what makes our University so special – from our distinguished history to the latest news and campus developments.
-
Colleges & Schools

Colleges & Schools
University of Galway has earned international recognition as a research-led university with a commitment to top quality teaching across a range of key areas of expertise.
-
Research & Innovation

Research & Innovation
University of Galway’s vibrant research community take on some of the most pressing challenges of our times.
-
Business & Industry

Guiding Breakthrough Research at University of Galway
We explore and facilitate commercial opportunities for the research community at University of Galway, as well as facilitating industry partnership.
-
Alumni & Friends

Alumni & Friends
There are 128,000 University of Galway alumni worldwide. Stay connected to your alumni community! Join our social networks and update your details online.
-
Community Engagement

Community Engagement
At University of Galway, we believe that the best learning takes place when you apply what you learn in a real world context. That's why many of our courses include work placements or community projects.
Biomedical Science (MSc)
MSc (Biomedical Science)
College of Science and Engineering, School of Biological and Chemical Sciences- Title of Award
- Master of Science
- Course Code
- MSC-MV1, MSC-MV4
- Average Intake
- 25
- Delivery
- On Campus
- NFQ
- Level 9
- Award Type
- Major
- Next Intake
- September 2026
- Duration
- 1 year, full-time | 2 years, full-time with industry placement
- ECTS Weighting
- 90
Why Choose This Course?
Course Information
Who is this course for?
The MSc in Biomedical science (90 ECTS) is a full-time Level 9 programme course, normally extending over a 12-month period from September to August. The MSc is intended to introduce students to science and engineering disciplines they would not have covered in their undergraduate studies.
- The course is designed for those who wish to follow careers as Biomedical Scientists in research, the Health Service or in the wider context of biomedical science including Medical Technologies, Bio-pharmaceutical and other Healthcare industries.
- Using state-of-the-art technologies and a range of skills from scientific, engineering and clinical disciplines to understand and investigate questions originating in biology and medicine.
- You will be introduced to science and engineering disciplines you will not have covered in your undergraduate studies. You will have access to some of Ireland’s leading researchers in this area.
- A major objective of the course is to introduce students to an interdisciplinary approach to Biomedical Science, which utilises technologies and skills from a wide spectrum of scientific, engineering and clinical disciplines.
What will I study?
The full-time Level 9 programme normally extends over a 12-month period from September to August. The MSc is intended to introduce students to science and engineering disciplines they would not have covered in their undergraduate studies.
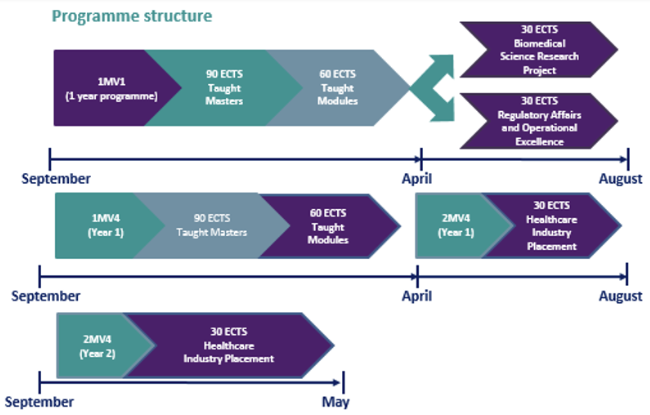
The first two semesters consist of taught modules (60 ECTS; see module details for more information) that will acquaint you with the basics of Biology, Chemistry and Physics, depending on your background. The modules are devoted to coursework through a series of lectures and tutorials and assessed by means of a written exam and/or continuous assessment. Students complete both scientific and business modules. Students are expected to carry out a business project by preparing a written report and presentation on a biomedical science-related topic. Also, during the first two semesters, there will be a series of practical sessions that will familiarise you with different forms of instrumentation and techniques.
An optional laboratory-based research project (30 ECTS) is undertaken by students in the 3rd semester (April–August). Toward the end of the research project, students are asked to present their findings at an MSc Research Symposium Day. There is a prize for the most outstanding MSc Research presentation on the day.
An alternative optional 30 ECTS module can be chosen instead by students. This module Regulatory Affairs & Operational Excellence aims to equip students with foundational practical knowledge of the regulatory pathways for placing medical devices and pharmaceuticals on the market within the EU, US and globally. It explains the legislation applicable and guidelines available to medical device and pharmaceutical manufacturers. Students also gain theoretical and practical knowledge and experience in Lean Six Sigma Operational Excellence, and Quality Management.
In a new development from 2018–19, students undertaking the MSc. also have the opportunity to apply for a 9–12-month industry placement, which if successfully secured, is carried out instead of the research dissertation. An industry placement-associated module (30 ECTS), is completed by students undertaking industry placements, replacing the research dissertation module. Students undertaking an industry placement will be registered on the MSc into a 2nd year for the duration of the placement.
1MV1
Semester 3: Research Project
A 4-month laboratory project with an academic research team on a subject related to biomedical science. The aim of this module is to provide students with hands-on experience of the rigours of scientific research, from experimental design to execution of research. Students are educated as to best practice for reporting their results.
Semester 3: Regulatory Affairs & Operational Excellence
The module provides MSc in Biomedical Science students with a detailed foundation in Regulatory Affairs (both EU & US), Lean Six Sigma Operational Excellence, and Quality Management. Students are introduced to world-leading device technologies for global disease treatment being manufactured, and new and emerging breakthrough technologies. It also provides an introduction to state-of-the-art theories and concepts in relation to a research topic in the Quality Management area.
1MV4
Semesters 3–5: Industry Placement
The Industry Placement module provides a period of professional work experience of up to 12 months in a Medical Technologies or Pharmacuetical/BioPharmaceutical company for MSc in Biomedical Science students. Students gain important on-the-job experience and skills through work placements with participating companies. Placements take place from June in the first year and run to end of May in Year 2. A placement introduces the student to structured employment and will develop an understanding of the organisation, its procedures, good manufacturing practice, technologies and its products. The industry placement provides students with a range of professional development opportunities, including:
- Valuable work experience in a relevant technical and business area.
- Develop a range of work-related technical, business, organisational, team and management skills demanded by graduate employers.
- Apply skills developed during the MSc. degree programme.
- Develop technical skills.
- Gain insights into future career opportunities and enable more informed career choices.
- Improve employability.
- Develop both personally and professionally: gaining confidence, working individually and in a team, taking on positions of responsibility, making decisions, enhancing communications, reporting skills, and supporting others.
- Gain experience in CV preparation and job application interview skills.
Curriculum Information
Curriculum information relates to the current academic year (in most cases).Course and module offerings and details may be subject to change.
Glossary of Terms
- Credits
- You must earn a defined number of credits (aka ECTS) to complete each year of your course. You do this by taking all of its required modules as well as the correct number of optional modules to obtain that year's total number of credits.
- Module
- An examinable portion of a subject or course, for which you attend lectures and/or tutorials and carry out assignments. E.g. Algebra and Calculus could be modules within the subject Mathematics. Each module has a unique module code eg. MA140.
- Subject
- Some courses allow you to choose subjects, where related modules are grouped together. Subjects have their own required number of credits, so you must take all that subject's required modules and may also need to obtain the remainder of the subject's total credits by choosing from its available optional modules.
- Optional
- A module you may choose to study.
- Required
- A module that you must study if you choose this course (or subject).
- Required Core Subject
- A subject you must study because it's integral to that course.
- Semester
- Most courses have 2 semesters (aka terms) per year, so a three-year course will have six semesters in total. For clarity, this page will refer to the first semester of year 2 as 'Semester 3'.
Year 1 (90 Credits)
OptionalAN230: Human Body Structure - 5 Credits - Semester 1OptionalSI317: Human Body Function - 10 Credits - Semester 1
OptionalPM208: Fundamental Concepts in Pharmacology - 5 Credits - Semester 1
OptionalPH339: Radiation and Medical Physics - 5 Credits - Semester 1
OptionalBI5107: Introduction to Molecular and Cellular Biology - 5 Credits - Semester 1
OptionalBES556: Research & Minor Thesis - 30 Credits - Semester 1
OptionalBES5121: Healthcare Technologies and Operations Innovations - 15 Credits - Semester 1
OptionalBI5110: Cancer Biology - 5 Credits - Semester 1
OptionalPM209: Applied Concepts in Pharmacology - 5 Credits - Semester 1
RequiredBES5119: Science Communication - 5 Credits - Semester 1
RequiredBES5107: Applied Biomedical Sciences - 10 Credits - Semester 1
RequiredBME405: Tissue Engineering - 5 Credits - Semester 1
RequiredMG529: Introduction To Business - 10 Credits - Semester 1
OptionalBG5104: Protein Technology - 5 Credits - Semester 2
OptionalBME502: Advanced Tissue Engineering - 5 Credits - Semester 2
OptionalBES5120: Healthcare Regulatory Affairs & Operational Excellence - 15 Credits - Semester 2
OptionalMA3103: Introduction to Bioinformatics - 5 Credits - Semester 2
RequiredPH5124: Materials Science & Biomaterials - 5 Credits - Semester 2
RequiredBES5104: Regulatory Compliance in Healthcare Manufacturing - 10 Credits - Semester 2
RequiredBES554: Molecular Medicine - 5 Credits - Semester 2
- Interdisciplinary approach: A major objective of the course is to introduce students to an interdisciplinary approach to Biomedical Science, which utilises technologies and skills from a wide spectrum of scientific, engineering and clinical disciplines.
- Research-led teaching: Ensures students learn from academics actively engaged in cutting-edged research, keeping course content current and relevant.
- Industry Placement: The programme is placed in the hub of the MedTech sector offering a 12-month industry placement opportunity. Offers real-world experiences through placements, enhancing employability and practical understanding of scientific careers within the biomedical science field.
- Develops Research Skills: Hand-on practical and laboratory experience training students in scientific methods, data collection, and evidence-based conclusions.
- Promotes critical thinking and problem-solving abilities: Encourages analytical and problem-solving skills through observation, experimentation, and data interpretation.
- Builds Communication Skills: Enhances ability to present scientific ideas clearly through reports, presentations and discussions.
With a focus on real-world application, industry-relevant modules, and opportunities for placements, this degree prepares you for a wide range of exciting careers in biomedical science.
Industry-Relevant Modules like Tissue Engineering and Applied Biomedical Sciences will give you the skills employers are looking for in today's economy.
Graduates have gone on to further research at PhD level and have also found employment with a range of companies, including Boston Scientific, Medtronic, Regeneron, Abbott, Allergan, and Pfizer, and many Medtech, and Pharma/Biopharma Healthcare industries.
Students undertaking the MSc. have the opportunity to apply for a 9–12-month industry placement, which if successfully secured, is carried out instead of the research dissertation.
The Industry Placement module provides a period of professional work experience of up to 12 months in a Medical Technologies or Pharmacuetical/BioPharmaceutical company for MSc in Biomedical Science students. Students gain important on-the-job experience and skills through work placements with participating companies. Placements take place from June in the first year and run to end of May in Year 2. A placement introduces the student to structured employment and will develop an understanding of the organisation, its procedures, good manufacturing practice, technologies and its products. The industry placement provides students with a range of professional development opportunities, including:
- Valuable work experience in a relevant technical and business area.
- Develop a range of work-related technical, business, organisational, team and management skills demanded by graduate employers.
- Apply skills developed during the MSc. degree programme.
- Develop technical skills.
- Gain insights into future career opportunities and enable more informed career choices.
- Improve employability.
- Develop both personally and professionally: gaining confidence, working individually and in a team, taking on positions of responsibility, making decisions, enhancing communications, reporting skills, and supporting others.
- Gain experience in CV preparation and job application interview skills.
The MSc in Biomedical Science course is not accredited by the Academy for Clinical Science and laboratory Medical Scientists (ACSML) or CORU. As a result, graduates of the MSc are not eligible to be employed as Laboratory Medical Scientists in Hospital clinical diagnostics laboratories.
Graduates have however, secured Research Assistant positions in laboratories within Hospital clinical units.
- Dr Mary Ní Fhlathartaigh
- Dr Yury Rochev
- Prof. Gerard Wall
- Dr Andrew Flaus
- Prof. Peter Dockery
- Prof. Eilís Dowd
- Dr Olivia Mc Dermott
- Dr Lars Jermiin
- Dr Manus Biggs
- Dr Florence Abram
- Dr Sheila Donnelly
- Dr Aidan Toner
- Dr Mark Foley
- Ms. Niamh Nolan
How will I learn?
The MSc in Biomedical Science combines innovative teaching methods with practical, hands-on learning, industry site visits and industry guest lectures to ensure a comprehensive educational experience. You will learn through a mix of interactive lectures, seminars and workshops led by expert faculty. Real-world case studies and data-driven projects will enable you to apply theoretical knowledge to practical problems.
Group projects and collaborative activities will enhance your teamwork and communication skills, while individual assignments and the final dissertation will help you develop independence and critical thinking.
How Will I Be Assessed?
Students are formally assessed through a range of continuous assessment and end-of-semester examinations in Semester 1. Continuous assessment involves written assignments, projects, presentations, video pitches and case studies. Written examinations include short answer examinations, multiple-choice-question examinations during the semester, and end of Semester 1 examinations in December.
Assessment of students in Semester 2 is mainly through continuous assessment of each module, through a combination of written assignments, evaluations of laboratory practical skills, laboratory reports, workshop-based problem solving, oral presentations.
The Research Project dissertation is also assessed. Assessment includes examination of the written research thesis, as well as oral and poster presentations, and assessment of student performance in conducting their research project.
una.canney@universityofgalway.ie
or
mary.nifhlathartaigh@universityofgalway.ie
Programme Director(s):
Dr Mary Ní Fhlathartaigh
E: mary.nifhlathartaigh@universityofgalway.ie
School of Biological and Chemical Sciences
College of Science and Engineering
Graduates with a Level 7 degree and who can demonstrate that they have at least 2-3 years relevant experience in research or the Medical Technology, Healthcare or Biotechnology industry .
Graduates of the MSc in Biomedical Science will be able to:
- Demonstrate a clear, consise understanding of the principles of the biomedical science field
- Evaluate received knowledge and articulate their own contribution to the existing scholarship
- Perform a wide range of laboratory techniques in biomedical science
- Design, conduct, and evaluate scientific investigations
- Apply enhanced critical thinking and analytical skills
- Work effectively as part of a multidisciplinary team while meeting deadlines and project goals
Accreditations & Awards
Meet our Employers
Entry Requirements and Fees
Minimum Entry Requirements
Candidates must hold at least a Second Class Honours Level 8 primary degree in a related biological or biomedical sciences, physical sciences or engineering subject area or hold a Level 7 degree and who can demonstrate that they have at least 2-3 years relevant experience in research or the Medical Technology, Healthcare or Biotechnology industry.
Academic entry requirements standardised per country are available here.
English Language Entry Requirements
For applicants whose first language is not English, an English language proficiency of IELTS score of 6.5 is required (with no less than 6.5 in Writing and no less than 6.0 in any other band) or equivalent.
More information on English language test equivalency are available here.
Supporting Documents
You will be required to provide supporting documentation as part of your application. You can check here what supporting documents are required for this course.
You can apply online to the University of Galway application portal here.
Please review the entry requirements set out in the section above.
You will be required to upload supporting documentation to your application electronically. See the section above on entry requirements for further information on the supporting documentation required for this course.
Closing Dates
For this programme, there is no specific closing date for receipt of applications. Applications will be accepted on a rolling basis and course quotes will be reviewed continuously throughout the application cycle.
Notes
- You will need an active email account to use the website and you'll be guided through the system, step by step, until you complete the online form.
- Browse the FAQ's section for further guidance.
Fees for Academic Year 2026/27
| Course Type | Year | EU Tuition | Student Contribution | Non-EU Tuition | Levy | Total Fee | Total EU Fee | Total Non-EU Fee |
|---|---|---|---|---|---|---|---|---|
| Masters Full Time | 1 | €8,900 | €28,500 | €140 | €9,040 | €28,640 |
For 26/27 entrants, where the course duration is greater than 1 year, there is an inflationary increase approved of 1.8% per annum for continuing years fees.
Postgraduate students in receipt of a SUSI grant – please note an F4 grant is where SUSI will pay €4,500 towards your tuition (2026/27). You will be liable for the remainder of the total fee. A P1 grant is where SUSI will pay tuition up to a maximum of €6,270. SUSI will not cover the student levy of €140.
Note to non-EU students: learn about the 24-month Stayback Visa here.
Postgraduate Excellence Scholarships
This scholarship is valued at €1,500 for EU students applying for full-time taught master's postgraduate courses. You will be eligible if:
- You have been accepted to a full-time taught master's course at University of Galway,
- You have attained a first class honours (or equivalent) in a Level 8 primary degree.
An application for the scholarship scheme is required (separate to the application for a place on the programme). The application portal for 2026 is now open and available here. Applications will close on the 30th September 2026. Full details available here.
Global Scholarships
University of Galway offers a range of merit-based scholarships to students from a number of countries outside of the EU. Visit here for schemes currently available.
School of Biological and Chemical Sciences Scholarships
The School of Biological and Chemical Sciences offers the following scholarships:
- One Excellence Scholarships of €10,000. The Programme Director will award this scholarship to the applicant with the highest overall application.
- €2,000 Merit Scholarships for all Outside of the EU students. This will be organised automatically by the School.
Application Process
Students applying for full time postgraduate programmes from outside of the European Union (EU), You can apply online to the University of Galway application portal here.
Our application portal opens on the 1st October each year for entry the following September.
Further Information
Please visit the postgraduate admissions webpage for further information on closing dates, documentation requirements, application fees and the application process.
Programme Director
Dr Mary Ní Fhlathartaigh
E: mary.nifhlathartaigh@universityofgalway.ie
Why University of Galway?
World renowned research led university nestled in the vibrant heart of Galway city on Ireland's scenic West Coast.
Downloads
Meet Our Alumni
Course Introduction
Underpinning knowledge and practical skills to pursue a successful career in biomedical science.
The course is designed for those who wish to follow careers as Biomedical Scientists in research, the Health Service or in the wider context of biomedical science (including Medical Technologies, Bio-pharmaceutical and other Healthcare industries). Using state of the art technologies and a range of skills from scientific, engineering and clinical disciplines to understand and investigate questions originating in biology and medicine. You will be introduced to science and engineering disciplines you will not have covered in your undergraduate studies. You will have access to some of Ireland’s leading researchers in this area.

















.jpg)












